Executive Summary (TL;DR)
FSMA 204 sets a new standard for traceability: data must be structured, linked, and delivered as a sortable electronic dataset within 24 hours.
Pet food may not be on the Food Traceability List, but that distinction does not hold in practice. Your suppliers already operate under FSMA 204, and retailers and auditors expect the same speed and structure from you. If your system cannot ingest, link, and retrieve that data instantly, traceability breaks at intake.
Most failures are not due to missing data, but disconnected systems. Spreadsheets hide recurring issues, CAPAs lack verification, and supplier risk is not tracked in real time. Under audit or recall conditions, this is treated as lack of control, not delay.
In 2026, the cost of slow traceability is measured in recall scope, lost shelf space, and damaged trust. Manual systems store data. They do not control it.
Pet food businesses now need systems that connect supplier data, traceability events, NCRs, CAPAs, and documents into a single, time-stamped chain, so issues can be isolated quickly and proven with confidence.
FSMA 204 for Pet Food: Why the "Loophole" is a Liability?
The Regulatory Paradox: FSMA 204, the Food Traceability Rule, introduces a clear mandate for structured, linked, and rapidly retrievable data. The FDA’s objective is to enable faster identification of contaminated products, with a strict expectation that records are delivered as an electronic, sortable dataset within 24 hours.
According to FDA FAQ TFTL.8, pet food and animal feed are not currently included on the Food Traceability List (FTL) and are therefore not subject to the rule's specific additional recordkeeping requirements. Furthermore, animal food made from FTL by-products is also technically exempt.
But for a Pet Food business, this "Loophole" is a trap. Here is the operational reality:
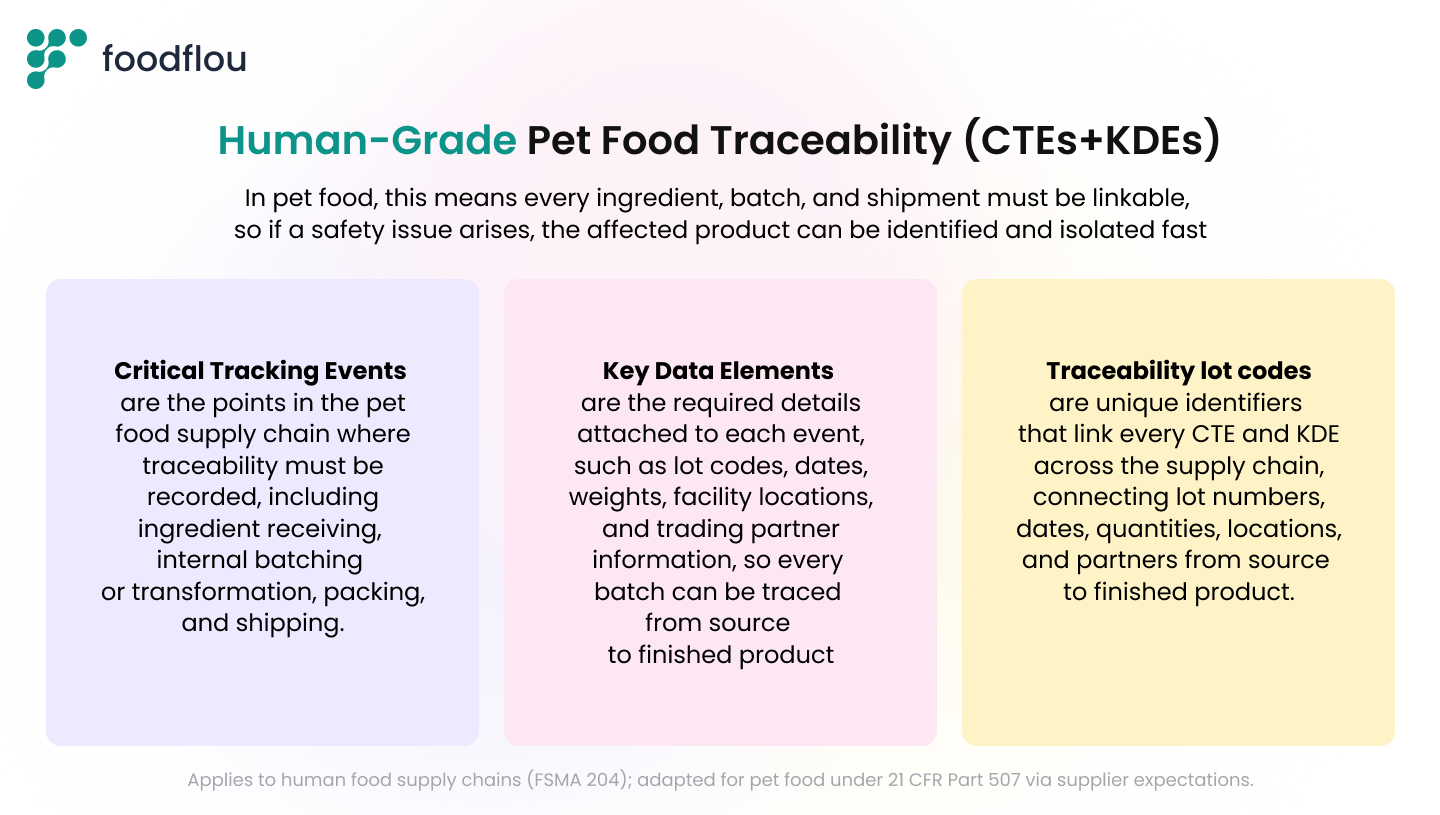
1. The FDA has standardized the audit vocabulary into two pillars:
- Critical Tracking Events (CTEs): these are the specific points where a product is handled or changes state, such as receiving raw materials, processing, packing, or shipping. Each event represents a moment where traceability must be captured to track the flow of product.
- Key Data Elements (KDEs): these are the required data points recorded at each event, including lot or batch identifiers, supplier and recipient details, dates, quantities, and locations. These elements must be consistently captured and linked so the full product journey can be reconstructed quickly and accurately.
The Goal: By linking KDEs to CTEs, you replace fragmented spreadsheets with a single, verifiable chain of data. Even if the FDA doesn't mandate KDEs for pet food yet, your upstream suppliers (proteins, fats, vitamins) are often in the human food system and must provide this digital data. If your system cannot ingest and link their 204-compliant files, your internal traceability breaks the moment the raw material arrives.
FSMA 204 Traceability Meets Pet Food Reality
The Misconception: Finished pet food isn’t explicitly on the Food Traceability List (FTL), leading many to a false sense of security.
The Reality: Pet food is still regulated as animal food under the Food Safety Modernization Act, specifically under Preventive Controls for Animal Food in 21 CFR Part 507. This requires manufacturers to conduct hazard analysis, implement preventive controls, manage supplier risk, and maintain records that demonstrate those controls are consistently applied.
What FSMA 204 changes is not the obligation itself, but the standard of proof. The rule introduces structured traceability requirements for foods on the Food Traceability List (FTL), including the expectation that traceability data is captured at defined events, linked across the supply chain, and provided in an electronic, sortable format within 24 hours when requested.
Why the "Exemption" is a Myth in Practice:
While finished pet food is not currently on the FDA’s Food Traceability List (FTL), the operational reality in 2026 makes this distinction irrelevant for four reasons:
- If you cannot produce a linked, sortable electronic record during an inspection, it is no longer treated as a clerical delay. It is cited on a Form 483 as a Significant Failure of Preventive Controls under 21 CFR Part 507.
- The market moves faster than the law. Platforms like Chewy and Petco do not differentiate between FTL and non-FTL products. They are increasingly making 24-hour digital traceability a contractual requirement. To these retailers, the FDA FAQ is a legal technicality; to their brand protection teams, a slow response is a reason to pause your listing.
- Most systems fail not because data is missing, but because it is disconnected. When a safety event occurs, every hour of delay forces a wider "defensive recall." You end up pulling safe product off shelves simply because you cannot prove its safety fast enough.
Bottom Line: From a regulatory standpoint, accuracy and accessibility are non-negotiable. During an FDA inspection, the ability to retrieve and present records efficiently is the only way to demonstrate you are in control of your supply chain. Relying on an exemption won't protect your shelf presence or your brand equity, only structure and speed will.
CAPA and NCR in Pet Food: Why Systems Break When Audits Start?
Most pet food companies have CAPA processes in place. The issue is not documentation. It is execution. Corrective and Preventive Actions under Preventive Controls for Animal Food must show that issues are identified, corrected, and prevented from repeating. That requires more than logging a deviation. It requires a clear, verifiable chain from root cause to effective resolution. In a manual system, this looks complete, but it fails the digital sortability test now expected in 2026. The gap appears when that process is tested:
- NCRs in spreadsheets hide recurring supplier or batch patterns
- CAPAs lack timestamps, ownership, or supporting evidence
- Closure is recorded without clear verification
- Recurring issues are not visible across time, suppliers, or production
The Regulatory Standard: Proof of Effectiveness
From a 2026 regulatory standpoint, handling an issue is only 20% of the requirement. To satisfy an auditor or a Form 483 inquiry, you must demonstrate a four-part "Chain of Control":
- Definitive Root Cause: Moving beyond "human error" to identify systemic triggers.
- Implementation Proof: Timestamped evidence that the corrective action was deployed.
- Verification of Effectiveness: Data showing the issue did not return after the fix.
- Preventative Logic: Evidence that the system was updated to block similar issues across other lines or facilities.
If your chain of evidence is incomplete, the auditors consider your CAPA ineffective, which signals a systemic lack of control over your food safety plan.
To bridge this gap, pet food brands must move to structured, linked workflows. This means capturing deviations at the source (the plant floor), assigning clear digital ownership, and automatically linking supporting evidence to every step. Without this structure, the same problems will inevitably reappear, and during an audit, a recurring issue is no longer an accident, it's a significant Non-Compliance.

Supplier Risk Management in Pet Food: From Periodic Checks to Continuous Control
Supplier approval is no longer a "one-and-done" checkbox. Under 21 CFR Part 507, the FDA has shifted the mandate from simple onboarding to continuous oversight. Pet food manufacturers are now legally required to ensure that suppliers meet safety standards not just on day one, but for every subsequent batch.
The primary failure point in 2026 is rarely a lack of data, it is a lack of control. Supplier risk is dynamic: certificates of insurance expire, audit outcomes fluctuate, and ingredient specs vary by harvest. When your system relies on manual checks, you are operating on "stale" compliance data. In a regulatory environment that demands real-time accuracy, outdated assumptions are indistinguishable from non-compliance.
In 2026, the stakes have shifted. Maize and protein suppliers are now under heavy scrutiny due to recent Mycotoxin (DON) and Salmonella trends. If your supplier is not providing digital KDEs for these high-risk ingredients, you are essentially inheriting their liability. When a supplier falls out of compliance and it is not flagged, that risk moves directly into your production. At that point, ownership is no longer external; it sits fully with the manufacturer.
Regulatory expectations are clear: your Approved Supplier List must be a live document. This requires a system that moves beyond storage and into active evaluation. To stay ahead of a Form 483, your QMS must provide:
- Real time tracking of document validity
- Supplier performance linked across NCRs, CAPAs, and incidents
- Early visibility of recurring issues
- Approved supplier lists that reflect live compliance status
The Operational Cost: Manual tracking does not just slow you down. A single audit can take 40- 200 hours of QA time, pulling senior teams away from production. This sits within a broader compliance burden of over $50k per employee each year, much of it spent on admin instead of risk control. That effort is absorbed by senior QA and operations teams pulling data together instead of managing risk.
Pet Food Traceability, Recalls, and the Cost of Delay
The primary objective of FSMA 204 is speed. In a safety event, speed determines how far the issue spreads and how expensive it becomes. Recent 2026 thiamine and bacterial advisories (e.g., Quest, Elite Treats) have proven that the FDA is no longer waiting for confirmation of wide-spread illness - they are issuing advisories based on a single "out of spec" test. At this speed, "missing data" is rarely the problem, the problem is the inability to define scope.
Without linked, batch-level visibility, companies fall into a financial cliff. You end up pulling 100,000 bags simply because you cannot prove which 5,000 actually used a specific lot of tainted protein or vitamin premix, and to a pet owner, there is no "small" safety issue. Once trust is questioned, the damage ripples through repeat purchases, retailer confidence, and long-term brand value.
When traceability is structured and immediate, the response changes completely:
- Affected batches are isolated quickly, down to specific lot codes rather than entire production runs
- Supplier issues are traced back to source before they carry into additional batches
- Corrective actions are focused on the actual failure point instead of disrupting wider operations
This is where batch level traceability stops being theoretical and starts driving outcomes. It determines whether an issue is contained early or allowed to spread across production, distribution, and customers. The 24-hour FSMA 204 requirement sets the regulatory minimum, but the market expects faster responses. Every hour of delay includes more product, affects more customers, and makes containment harder. Manual systems store information. Structured platforms let you act on it immediately.
This is exactly the gap foodflou is built to close. Not storing records, but connecting them into a system that holds under pressure.
From Audit Preparation to Audit-Ready QMS: The New Pet Food Standard
The era of preparing for audits in the pet food industry is over. In 2026, the expectation from the FDA, GFSI standards such as SQF and BRCGS, and major retail partners is clear. Your system must be audit ready at all times.
Auditors are no longer reviewing a curated sample of records. They are testing the integrity of your operation. They follow the links between supplier COAs, ingredient intake, internal batching, and CAPA verification to see if your food safety system holds together under real conditions.
In pet food, this standard is higher because the risk profile is more complex and more regulated across multiple layers. In addition to FDA requirements under FSMA and 21 CFR Part 507, manufacturers must align with AAFCO guidelines for nutritional adequacy, ingredient definitions, and labeling claims. What is declared on the label must be supported by verifiable data across sourcing, formulation, and production.
This is where audits go deep:
- Co-manufacturing and tolling require clear lot segregation and dual traceability across facilities
- Kill step validation must demonstrate consistent pathogen control, especially for Salmonella
- Allergen cross contact must be managed across multi run production
- Supplier and ingredient compliance must be current, not assumed
- Retail partners require proof, not statements
This is not about documentation. It is about control. At this level, the difference is operational. Manual systems rely on effort. They require teams to assemble, verify, and explain data every time it is requested. That introduces variability, delays, and risk. Structured systems remove that dependency. Data is already linked, current, and ready to use across traceability, supplier management, and CAPA workflows, and that difference shows up immediately when the system is tested. Not in theory. In response time, accuracy, and confidence. Preparing for audits becomes unnecessary because the system is already aligned with how the business operates.
And that is the shift. From proving compliance after the fact to demonstrating control in real time. The comparison below shows how that difference plays out across pet food traceability, supplier control, NCR and CAPA, and overall audit readiness.
Emerging Pressure from Consumers and Retailers
Regulation sets the floor. Consumers and retailers are raising the ceiling. Pet owners are not casual buyers anymore, around 67% research ingredients before purchasing, and demand for human grade pet food claims has grown sharply in recent years. People are applying the same standards to pet food as they do to their own meals. If the label says human grade, responsibly sourced, or clean, they expect that claim to hold up when questioned.
Retailers are aligning with that behavior. Requirements are becoming more specific and more immediate. Platforms like Chewy and Petco are pushing suppliers toward faster, more detailed pet food traceability, including lot level sourcing proof and response times measured in hours, not days.
This is where things get interesting. The questions coming from retailers and consumers are starting to mirror regulatory expectations. When someone asks where a protein comes from, they are effectively asking for the same linked data that sits behind FSMA traceability requirements. Supplier to batch, batch to product, all connected and verifiable.
The difference is that this pressure is not scheduled. It does not come with notice. And it carries real consequences.
Recent Salmonella recalls (95% of 2025's 13 pet food events) crushed brands with weak traceability. RCL Foods lost massive shelf presence and cancelled promotions through June 2026. One poor or delayed response can increase customer churn risk by up to 3x, while brands that respond quickly and clearly retain trust at significantly higher rates.
That puts traceability in pet food in a very different position.
It is no longer just supporting compliance or audits. It sits directly behind brand protection. It determines how confidently a business can respond, how quickly issues can be clarified, and whether customers and retailers continue to trust what is on the label. For pet food brands, especially those positioning around human grade pet food, ingredient quality, or sourcing transparency, this is not optional. If you make the claim, you need to prove it, quickly and clearly.
And that proof depends entirely on how well your data is structured, connected, and ready to use when the question comes.

What This Signals for Pet Food Brands
FSMA 204 demands 24hr traceability. 21 CFR Part 507 requires supplier control + CAPA proof. Audits, Chewy, pet parents expect answers now.
This is where most operations fail. Manual systems can store data, but they break when that data needs to be connected, verified, and delivered under pressure. That delay expands recall scope, increases cost, and exposes gaps in control.
foodflou is a food safety software built around that requirement. Supplier data, traceability events, NCRs, CAPAs, and documents are linked into a single, time stamped system. Certificates are tracked continuously, and issues are connected across suppliers, batches, and time.
The difference is simple. You either isolate an issue quickly, or you widen it across batches, customers, and revenue.
In 2026, the gap between 'exempt' and 'compliant' is the gap between a 2-hour data request and a 2-week recall. Which one is your team prepared for?Book a demo and see how fast you can respond when everything is already connected.




.svg)

.svg)







.png)




.svg)



.png)



.svg)

